TIGAR
The core phosphatase fold is
conserved between two proteins. As observed in PhoE and TIGAR
comparison,β6 and β7 strands are longer in TIGAR
compared to Fru-2,6-bisphosphatase(FBPase-2). In the active
region,the TIGAR α6 is replaced by two separate helices; and
loop region including α9 is not observed in FBPase-2. The
amino acids coordinating P1 and P2 in active region is shown
below.
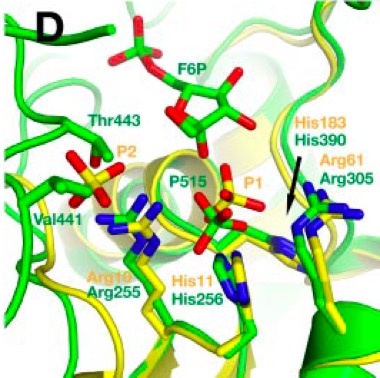
Phosphate ions bound are shown in space-filling view. Described helices and beta sheets are labeled. In superimposition picture above, TIGAR is shown in yellow and FBPase-2 is shown in green.
Hua Li and Gerwald Jogl
Structural and Biochemical Studies of TIGAR (TP53-induced Glycolysis and Apoptosis Regulator
The Journal of Biological Chemistry, 284,1748-1754
(PubMed)
3E9D (PDB)
The
two separate helices occupying the position of TIGAR α6 are
shown in green. In
contrast to TIGAR, there is only one binding site for phosphate
ion and binding site for second phosphate is hindered by Val441
and Thr443 side chains. Bound phosphate molecule in FBPase-2 is
slightly shifted towards His256 due to its shifted position
compared to His11 of TIGAR. FBPase-2 has additional 25 residues
in its C-terminus forming extended loop structures. These loops
enclose active site giving restricted access to it and
specificity for Fru-2,6-P2. Compared to this TIGAR has more
open, accessible active site. Overall, the active site is not
fully conserved between two enzymes.
Phosphate
ion bound is shown in space-filling view and
Fru-6-phosphate, Val441 and Thr443 are shown in sticks view.
Described helices are shown in green.
Yuen MH, Mizuguchi H,Lee YH, Cook PF, Uyeda K,Hasemann CA
Crystal structure of the H256A mutant of rat testis
fructose-6-phosphate,2-kinase/fructose-2,6-bisphosphatase.
Fructose 6-phosphate in the active site leads to mechanisms for
both mutant and wild type bisphosphatase activities.
Journal of Biological Chemistry, 274, 2176-2184
(PubMed)
2BIF (PDB)